Critique 265:Alcohol consumption and breast cancer prognosis after breast cancer diagnosis: a systematic review and meta‑analysis of the Japanese Breast Cancer Society Clinical Practice Guideline, 2022 edition
Authors
Nomura T, Kawai M, Fukuma Y, Koike Y, Ozaki S, Iwasaki M, Yamamoto S, Takamatsu K, Okamura H, Arai M, Ootani S, Iwata H, Saji S. Breast Cancer. 2023. https://doi.org/10.1007/s12282-023-01455-4
Author’s abstract
Alcohol consumption is internationally recognized as one of the compelling risk factors for breast cancer, but it does not necessarily correlate with the prognosis of breast cancer patients. Alcohol consumption in breast cancer patients was addressed in the 2022 Breast Cancer Clinical Practice Guidelines. A systematic review and meta-analysis of epidemiological studies on alcohol consumption and breast cancer recurrence, breast cancer-related mortality, all-cause mortality, and cardiovascular disease mortality in breast cancer patients was performed. The PubMed, Cochrane Library, and Ichushi-Web databases were searched for relevant publications reporting cohort or case–control studies published until March 2021. A total of 33 studies (32 cohort studies and 1 case–control study) met the eligibility criteria; 4638 cases of recurrence, 12,209 cases of breast cancer-specific mortality, and 21,945 cases of all-cause mortality were observed. With regard to breast cancer recurrence, 7 studies assessed pre-diagnosis alcohol consumption (relative risk (RR) 1.02, 95% confidence interval (95% CI) 0.77–1.37, p = 0.88) and 3 studies assessed post-diagnosis alcohol consumption (RR 0.96, 95% CI 0.85–1.10, p = 0.57), and no significant increase or decrease in risk was observed. With regard to breast cancer-related mortality, 19 studies assessed pre-diagnosis alcohol consumption (RR 1.02, 95% CI 0.93–1.11, p = 0.69), 9 studies assessed post-diagnosis alcohol consumption (RR 0.96, 95% CI 0.77–1.19, p = 0.70), and no significant increase or decrease in risk was observed. With regard to all-cause mortality, 18 studies assessed pre-diagnosis alcohol consumption (RR 0.90, 95% CI 0.82–0.99, p = 0.02), 8 studies assessed post-diagnosis alcohol consumption (RR 0.88, 95% CI 0.74–1.02, p = 0.08), and pre-diagnosis alcohol consumption was associated with a significantly decreased risk. With regard to cardiovascular disease mortality and alcohol consumption, 2 studies assessed it, and the RR was 0.47 (95% CI 0.28–0.79, p = 0.005), showing that alcohol consumption was associated with a significantly decreased risk. The limitations of this study are that drinking status was mainly based on a questionnaire survey, which is somewhat inaccurate and has many confounding factors, and the cut-off value for the maximum alcohol intake in many studies was low, and it is possible that the actual intake was only an appropriate amount. In many countries, a standard drinking amount is set, and wise decisions are required.
Forum comments
Background including previous results
ISFAR has previously reviewed 19 papers on associations between alcohol consumption and incidence of breast cancer, which is the most commonly occurring cancer in women and the most common cancer overall. There were more than 2.26 million new cases of breast cancer in women in 2020 (https://www.wcrf.org/cancer-trends/breast-cancer-statistics/).
Alcohol consumption is recognized as one of the risk factors contributing to breast cancer incidence. This association has been observed for both pre- and post-menopausal women of all ages, and is observed to be independent of the type of alcoholic beverage consumed (Rosenberg et al. 1993, Longnecker 1994, Hamajima et al. 2003, Petri et al. 2004, Key et al. 2006). The association with may also be more significant for certain subtypes of breast cancer for example, hormone-sensitive rather than hormone-insensitive subtypes (Millikan et al. 2008, Dolle et al. 2009, Kwan et al. 2009, Li et al. 2010, Kabat et al. 2011).
The increased risk of breast cancer from alcohol consumption is additive to the other risk factors. The other risk factors are genetic, environmental including age of menarche, childbearing, breastfeeding and hormone replacement therapy, and lifestyle including diet and exercise; these contribute more heavily to the overall breast cancer risk.
Much attention has been focused on alcohol consumption, however, since it is a modifiable lifestyle factor and may, therefore, be more easily ‘managed’ and ‘changed’ to reduce the risk for this cancer.
The association between alcohol consumption and incidence of breast cancer has long been a topic of research and initially yielded contradictory results. The first purported positive association between alcohol consumption and breast cancer was reported in 1977 by Williams and Horn. The results showed strong evidence of a dose-response relation; however, the slope of the dose-response curve was quite modest.
A positive linear association between alcohol consumption and breast cancer incidence has since been confirmed after pooling most of the large epidemiological studies. Indeed, the first meta-analyses indicated an additional increased risk of approximately 10% for every 10 g of alcohol consumed per day (Longnecker et al., 1988, 1994, Smith-Warner et al., 1998) compared with non-drinkers. Approximately 1 in 8 women in the USA and Europe develop breast cancer, that is, women have 12.5% risk of developing breast cancer. Consequently a 5 – 10% increase from alcohol consumption suggests that a woman’s total risk increases from 12.5% to 13.1 – 13.8% if she is a drinker compared to a non-drinker (Hendriks, 2020).
A recent overall extensive meta-analysis took 15 different factors into account for breast cancer risk and attempted to put them into context (Poorolajal et al., 2021). Estrogen/progesterone intake and late pregnancy were identified as the first and second most significant positive risk factors for breast cancer, respectively, whereas sufficient fruit/vegetable consumption and sufficient physical activity were the first and second most protective factors against breast cancer, respectively (see figure below). Alcohol consumption was ranked 10 out of the 11 positive risk factors.
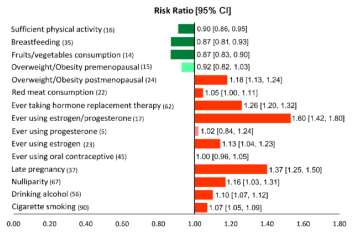
The role of the estrogen receptor (ER) in the association between alcohol consumption and breast cancer risk is also an interesting debate. Some authors report that the effect of drinking on the incidence of breast cancer is mainly manifested in estrogen receptor positive breast cancers (about 10% per any additional 10 g/day) (Sun et al., 2020).
Also, evidence for an association of alcohol consumption with prognosis after a diagnosis of breast cancer has been inconsistent. Based on a meta-analysis of 11 studies Ali et al. (2014) concluded that there was little evidence that pre- or post-diagnosis alcohol consumption is associated with breast cancer-specific mortality for women with ER-positive disease. There was weak evidence that moderate post-diagnosis alcohol intake is associated with a small reduction in breast cancer-specific mortality in ER-negative disease.
Forum Comments
This paper by Nomura et al. (2023) reports on a quantitative and qualitative systematic review and meta-analysis performed according to the Minds Handbook for Clinical Practice Guideline Development, a guideline issued by the Japanese Council for Quality Health Care. The paper was published in the Breast Cancer Society´s journal called Breast Cancer as a follow-up of questions raised while issuing their clinical practice guidelines, namely ‘Is alcohol consumption associated with prognosis in breast cancer patients?’.
Methodology
Papers were selected from international literature using relevant original papers and moderate quality review papers according to AMSTAR. Selection yielded 33 studies, 32 of which were cohort studies, in total covering 4,638 cases of recurring breast cancer. Follow-up varied between about 3 and 11 years.
Interestingly, this study not only evaluated the recurrence of breast cancer in association with alcohol consumption. The authors also assessed some benefits associated with alcohol consumption. This was done because the reduction in all-cause mortality associated with alcohol consumption after the diagnosis of breast cancer may be related to a decreased cardiovascular disease mortality. So, papers were included in this meta-analysis when alcohol consumption was not only studied in relation to breast cancer recurrence, but also in relation to breast cancer-related mortality, all-cause mortality in breast cancer patients and cardiovascular disease mortality in both breast cancer patients and healthy patients. This meta-analysis, therefore, also reported on 12,209 cases of breast cancer related mortality, 21,945 cases of all-cause mortality and 7,727 cases of cardiovascular mortality. An advantage of such an approach is that linkage is made with existing and well-described associations on alcohol consumption and health outcomes. This may further strengthen the reliability of the results obtained in this relatively new area of meta-analysis.
A full overview of potential risk of bias was provided in the paper. Most studies were unclear on the possible risk of selection bias of the non-exposed cohort, mainly because these studies were observational and therefore not randomly assigned. But the authors considered the risk for allocation concealment, blinding of participants and personnel as well as selective reporting low. A minority of studies had an unclear risk of incomplete outcome data mainly caused by a short follow-up period. Similarly, adjustment for confounding was inadequate 5 studies included and insufficient in an additional 7 out of the 33 studies included. This issue was mentioned as a limitation of this meta-analysis but were considered not to affect the overall conclusions of the meta-analysis, since the recommendation was in line with the outcomes reported for all 33 studies included.
Results
The main result indicates that all studies combined do not show an increased nor a decreased risk for breast cancer recurrence associated with alcohol consumption. This conclusion is surprising since alcohol has been positively associated with breast cancer risk in various meta-analyses. There is not a priori reason to assume that the risk for breast cancer may change after breast cancer has occurred in these women. Therefore, one would expect breast cancer risk attributable to alcohol consumption to be similar both before and after breast cancer diagnosis. One of the possible explanations, which was not discussed by the authors, may be that the studies selected for this meta-analysis were studies having included women drinking lightly or only moderately. Exposure in most studies varied from < 4 g/d up to 1-2 drinks per day. Only a few studies included concerned heavier drinking women, which may have beneficially affected the outcome.
The studies analysed numerous variables that may affect outcome. Firstly, the authors analysed the effect of pre- and post-diagnosis assessment of alcohol consumption in relation to the breast cancer recurrence. Both the analyses did not show a significant association. Also, the association of alcohol consumption with breast cancer mortality was investigated, both using pre- and postdiagnosis alcohol intake assessment and both for ER-positive and ER-negative cases. All associations were not significant with the ER-positive breast cancers having a borderline lower RR associated with alcohol consumption. A similar analysis on alcohol consumption and all-cause mortality in these cohorts showed that pre-diagnosis alcohol assessment was associated with a reduced RR, whereas post-diagnosis alcohol assessment was not associated with a changed RR. Breast cancer patients showed a small decrease in all-cause mortality risk, the reduced risk being present in both ER positive as well as in ER negative cases. These results correspond nicely with what is usually seen in general populations in which all participants are apparently healthy at the start of the study.
The authors concluded that these findings resulted in the following recommendation to the Japanese Breast Cancer Society Clinical Practice Guideline Committee: whether pre-diagnosis or post-diagnosis, alcohol consumption has a substantial effect on risk and is unlikely to increase the risk of breast cancer recurrence and death from breast cancer.
Discussion
Pre- and post-diagnosis alcohol consumption was analysed extensively in this study. The authors do not specify a reason for this specific interest, but it is logical to assume that women may change their drinking habits after breast cancer diagnosis. It is unclear from the study results if women did really change their drinking habits or not.
Another factor relevant to their guidance is the low occurrence of breast cancer in Japan, actually Japan has one of the lowest incidences of this type of cancer in the world. Although Japanese women have a relatively low risk of breast cancer compared with women in Western countries, the incidence rate has been increasing rapidly, and breast cancer is currently the leading cancer among Japanese women. The incidence rate of breast cancer among Japanese women increases with increasing age but decreases or flattens after 50 years of age, whereas it increases irrespective of age among women in Western countries (Liu et al., 2019). Japan also is one of the countries that has the highest life expectancy. One of the reasons may be related to the diet which is high in isoflavone, which is known to be associated with lower risk of breast cancer and prostate cancer. The relatively higher soy intake may account for the low breast and prostate cancer mortality in Japan. Soy and isoflavone intake have also been inversely associated with risk of cardiovascular diseases, especially cerebral and myocardial infarctions (Tsugane, 2020). It may have been interesting to see if studies performed in Japan and/or in that region of the world would have yielded similar results as compared to those in the rest of the world.
Also, the authors did not discuss the mechanism that may underly the absence of an increased risk of breast cancer reoccurrence. One may have expected a similar outcome, viz a similar positive association between alcohol consumption and breast cancer risk both before and after breast cancer diagnosis. The prevailing theory for the mechanism for breast cancer induction by alcohol consumption is that toxic metabolites of alcohol, namely acetaldehyde, would induce DNA mutations causing breast cancer. However, it is unclear whether acetaldehyde may be the real cause of breast cancer. It generally requires high levels of alcohol use and/or a deficiency in the ability to metabolize acetaldehyde. To what extent acetaldehyde accumulation occurs in breast tissue may not be immediately obvious, since ethanol metabolism is predominantly hepatic.
More indirect consequences of alcohol use, on oestrogen levels, or the effects of oestrogen, are another set of pathways by which alcohol could act more broadly, including in breast tissue. Thus, a broader perspective may be needed. Epigenetic mechanisms are likely to be affected through other mechanisms than oxidative ethanol metabolism and may be altered in multiple tissues. However, there is little specific understanding of the impact alcohol has on these regulatory mechanisms and, in particular there is no information on dose dependency and temporal consequences of these actions. Despite these insights, however, the overall lack of mechanistic understanding of these regulatory events is a major obstacle in understanding causal factors relevant for this disease (Zakhari & Hoek, 2018).
Specific Comments from Forum Members
Forum Member Ellison considers that “breast cancer is common around the world and appears to be the disease that is especially feared by most women. While diet, exercise, and obesity have been shown to affect the risk of breast cancer, the discovery that alcohol intake may be a factor in the initial diagnosis created an unusual amount of attention: finally, something that relates to the risk and can be avoided may lower the risk of this dread disease!
While most studies continue to show a slight increase in the risk of incident breast cancer from moderate drinking, the apparent degree of associated risk is usually seen to be very small (and alcohol intake is ranked very low in the list of potential risk factors, as discussed in this critique). Further, the risk of mortality from breast cancer has not been found to be increased by moderate alcohol intake, and total mortality is almost always found to be reduced by moderate drinking. Further, this study shows that alcohol consumption following the diagnosis of breast cancer may actually lower, rather than increase, the risk of total mortality.
Unfortunately, we remain mainly ignorant of important, modifiable steps that women can take to reduce their risk of breast cancer. In terms of the prevention of such cancer, avoiding moderate alcohol consumption is not the key factor we have been looking for.”
Forum Member Waterhouse stated that “in comparing this report to the famous Longnecker et al. (1988) study I am struck by the fact that all the studies cited here in Nomura et al. (2023) were published after 1988. There were three studies that predated 2000, but the majority were published after 2010. The evolution of research standards would suggest that the later studies were conducted with more stringent management, so the current meta-analysis should have a better foundation. It is interesting that Longnecker et al. (1988) clearly showed risk at higher rates of consumption, but mixed results at moderate rates, depending on the model, and this report clearly shows no elevated risk at moderate consumption levels. In the absence of evidence to the contrary, we should continue to discourage heavy consumption because it is likely to be associated with an increased risk of breast cancer, not to mention many other risks. This report, however, should relieve moderate drinkers of the fear of increasing their risk of breast cancer, and confirm that moderate consumption reduces overall mortality risk.
Forum Member Skovenborg adds that “this is a competent and non-biased review and meta-analysis showing that light alcohol consumption is not associated with breast cancer recurrence and breast cancer mortality, and actually it is associated with a reduced all-cause mortality. Among the seven lifestyle recommendations from the American Institute for Cancer Research and American Cancer Society is #(6) avoid alcohol. A recent prospective cohort study of 1340 patients with high-risk breast cancer found that high adherence to these recommendations was associated with a 37% reduced hazard of breast cancer recurrence and a 58% reduced hazard of mortality. (Cannioto et al. 2023). However, the benefits were due to full adherence to physical activity advice and non-smoking and recommendations regarding fruit and vegetable and sugar-sweetened beverage, while no statistically significant associations were observed for alcohol consumption and breast cancer recurrence and breast cancer mortality. Thus, the data from the cohort study by Cannioto et al. are in line with the meta-analysis from Nomura et al. (2023).”
Further, Forum Member Gaetano suggests that “even if moderate alcohol would (slightly) increase breast cancer risk, the greater reduction of CVD in women would remain a prevailing factor to reduce total mortality”.
References
Ali, A. M. G., Schmidt, M. K., Bolla, M. K., Wang, Q., Gago-Dominguez, M., Esteban Castelao, J., Carracedo, A., Garzón, V. M., Bojesen, S. E., Nordestgaard, B. G., Flyger, H., Chang-Claude, J., Vrieling, A., Rudolph, A., Seibold, P., Nevanlinna, H., Muranen, T. A., Aaltonen, K., Blomqvist, C., … Pharoah, P. D. (2014). Alcohol consumption and survival after a breast cancer diagnosis: a literature-based meta-analysis and collaborative analysis of data for 29,239 cases. Cancer Epidemiology, Biomarkers & Prevention : A Publication of the American Association for Cancer Research, Cosponsored by the American Society of Preventive Oncology, 23(6), 934–945. https://doi.org/10.1158/1055-9965.EPI-13-0901
Bagnardi, V., Rota, M., Botteri, E., Tramacere, I., Islami, F., Fedirko, V., Scotti, L., Jenab, M., Turati, F., Pasquali, E., Pelucchi, C., Bellocco, R., Negri, E., Corrao, G., Rehm, J., Boffetta, P., & La Vecchia, C. (2013). Light alcohol drinking and cancer: a meta-analysis. Annals of Oncology : Official Journal of the European Society for Medical Oncology, 24(2), 301–308. https://doi.org/10.1093/ANNONC/MDS337
Dolle, J.M., Daling, J.R., White, E., Brinton, L.A., Doody, D.R., Porter, P.L., Malone, K.E. (2009). Risk factors for triple negative breast cancer in women under the age of 45 years. Cancer Epidemiology Biomarkers and Prevention, 18, 1157–1166.
Hamajima, N., Hirose, K., Tajima, K., Rohan, T., Calle, E.E. et al. (2002). Collaborative Group on Hormonal Factors in Breast Cancer. Alcohol, tobacco and breast cancer–collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. British Journal of Cancer, 87(11), 1234-1245.
Hendriks, H. F. J. (2020). Alcohol and Human Health: What Is the Evidence? Annual Review of Food Science and Technology, 11, 1–21. https://doi.org/10.1146/ANNUREV-FOOD-032519-051827
Kabat, G.C., Kim, M., Phipps, A.I., Li, C.I., Messina, C.R., Wactawski-Wende, J., Kuller, L., Simon, M.S., Yasmeen, S., Wassertheil-Smoller, S., Rohan, T.E. (2011) Smoking and alcohol consumption in relation to risk of triple-negative breast cancer in a cohort of postmenopausal women. Cancer Causes and Control, 22(5), 775-783.
Key, J., Hodgson, S., Omar, R.Z., Jensen, T.K., Thompson, S.G., Boobis, A.R., Davies, D.S., Elliott, P. (2006). Meta-analysis of studies of alcohol and breast cancer with consideration of the methodological issues. Cancer Causes and Control, 17(6), 759-770.
Kwan, M.L., Kushi, L.H., Weltzien, E., Maring, B., Kutner, S.E., Fulton, R.S., Lee, M.M., Ambrosone, C.B., Caan, B.J. (2009). Epidemiology of breast cancer subtypes in two prospective cohort studies of breast cancer survivors. Breast Cancer Research, 11(3), R31.
Li, C.I., Chlebowski, R.T., Freiberg, M., Johnson, K.C., Kuller, L., Lane, D., Lessin, L., O’Sullivan, M.J., Wactawski-Wende, J., Yasmeen, S., Prentice, R. (2010) Alcohol consumption and risk of postmenopausal breast cancer by subtype: the women’s health initiative observational study. Journal of the National Cancer Institute, 102(18), 1422-1431.
Liu, R., Kitamura, Y., Kitamura, T., Sobue, T., Sado, J., Sugawara, Y., Matsuo, K., Nakayama, T., Tsuji, I., Ito, H., Suzuki, T., Katanoda, K., Tominaga, S. (2019). Reproductive and lifestyle factors related to breast cancer among Japanese women: An observational cohort study. https://doi.org/10.1097/MD.0000000000018315
Longnecker, M.P., Berlin, J.A., Orza, M.J., Chalmers, T.C. (1988) A meta-analysis of alcohol consumption in relation to risk of breast cancer. Journal of the American Medical Association, 260(5), 652-6. PMID: 3392790.
Longnecker, M.P. (1994) Alcoholic beverage consumption in relation to risk of breast cancer: meta-analysis and review. Cancer Causes and Control, 5(1), 73-82. doi: 10.1007/BF01830729. PMID: 8123780.
Millikan, R.C., Newman, B., Tse, C-K. (2008). Epidemiology of basal-like breast cancer. Breast Cancer Research on Treatment, 109, 123-139.
Nomura, T., Kawai, M., Fukuma, Y., Koike, Y., Ozaki, S., Iwasaki, M., Yamamoto, S., Takamatsu, K., Okamura, H., Arai, M., Ootani, S., Iwata, H., Saji, S. (2023). Alcohol consumption and breast cancer prognosis after breast cancer diagnosis: a systematic review and meta‑analysis of the Japanese Breast Cancer Society Clinical Practice Guideline, 2022 edition. Breast Cancer. https://doi.org/10.1007/s12282-023-01455-4.
Petri, A.L., Tjønneland, A., Gamborg, M., Johansen, D., Høidrup, S., Sørensen, T.I., Grønbaek, M. (2004). Alcohol intake, type of beverage, and risk of breast cancer in pre- and postmenopausal women. Alcoholism: Clinical and Experimental Research, 28(7), 1084-1090.
Poorolajal, J., Heidarimoghis, F., Karami, M., Cheraghi, Z., Gohari-Ensaf, F., Shahbazi, F., Zareie, B., Ameri, P., Sahraei, F. (2021). Factors for the primary prevention of breast cancer: A meta-analysis of prospective cohort studies. In Journal of Research in Health Sciences (Vol. 21, Issue 3, p. e00520). Hamadan University of Medical Sciences. https://doi.org/10.34172/jrhs.2021.57
Rosenberg, L., Metzger, L.S. Palmer, J.R. (1993) Alcohol consumption and risk of breast cancer: a review of the epidemiological evidence. Epidemiology Reviews, 15, 133–144.
Smith-Warner, S. A., Spiegelman, D., Yaun, S. S., Van Den Brandt, P. A., Folsom, A. R., Goldbohm, R. A., Graham, S., Holmberg, L., Howe, G. R., Marshall, J. R., Miller, A. B., Potter, J. D., Speizer, F. E., Willett, W. C., Wolk, A., Hunter, D. J. (1998). Alcohol and breast cancer in women: a pooled analysis of cohort studies. Journal of the American Medical Association, 279(7), 535–540. https://doi.org/10.1001/JAMA.279.7.535
Sun, Q., Xie, W., Wang, Y., Chong, F., Song, M., Li, T., Xu, L., Song, C. (2020). Alcohol consumption by beverage type and risk of breast cancer: A dose-response meta-analysis of prospective cohort studies. Alcohol and Alcoholism, 55(3), pp. 246–253. https://doi.org/10.1093/alcalc/agaa012
Tsugane, S. (2020). Why has Japan become the world’s most long-lived country: insights from a food and nutrition perspective. European Journal of Clinical Nutrition 75(6), 921–928. https://doi.org/10.1038/s41430-020-0677-5
Zakhari, S., Hoek, J. B. (2018). Epidemiology of moderate alcohol consumption and breast cancer: Association or causation? Cancers, 10, (10). https://doi.org/10.3390/cancers10100349
Comments on this critique by the International Scientific Forum on Alcohol Research were provided by the following members:
Henk Hendriks, PhD, Netherlands
Creina Stockley, PhD, MBA, Adjunct Senior Lecturer in the School of Agriculture, Food and Wine at the University of Adelaide, Australia
R Curtis Ellison, MD, Section of Preventive Medicine/Epidemiology, Boston University School of Medicine, Boston, MA, USA
Andrew Waterhouse, PhD, Department of Viticulture and Enology, University of California, Davis.
Erik Skovenborg, MD, specialized in family medicine, member of the Scandinavian Medical Alcohol Board, Aarhus, Denmark
Pierre-Louis Teissedre, PhD, Faculty of Oenology–ISVV, University Victor Segalen Bordeaux 2, Bordeaux, France
Giovanni Gaetano, MD, PhD, Department of Epidemiology and Prevention, IRCCS Istituto Neurologico Mediterraneo NEUROMED, Pozzilli, Italy
